Category: Medications - Page 3

Understand how deductibles, copays, and coinsurance work for prescription drugs. Learn what you pay out-of-pocket, how to track costs, and how to avoid surprise bills on your medications.

Authorized generics are identical to brand-name drugs, while traditional generics may differ in inactive ingredients. Know which one you're getting-and why it matters for your health.

The ANDA process is the legal pathway for generic drug approval in the U.S., requiring strict proof of bioequivalence, identical formulation, and cGMP compliance under the Hatch-Waxman Act. Learn the key requirements, fees, and common pitfalls.

Insurers push for generic drugs to cut costs, but providers are fighting back against rigid substitution rules that ignore patient safety. Learn how doctors navigate prior auth, state laws, and electronic systems to protect their patients.

Learn how to check formularies and preferred drug lists before prescribing to avoid coverage denials, high out-of-pocket costs, and treatment delays for your patients. Essential for doctors in 2025.

Learn how to store medications properly to prevent early expiration, avoid health risks, and save money. Discover the top storage mistakes, what each type of drug needs, and how to spot when meds have gone bad.
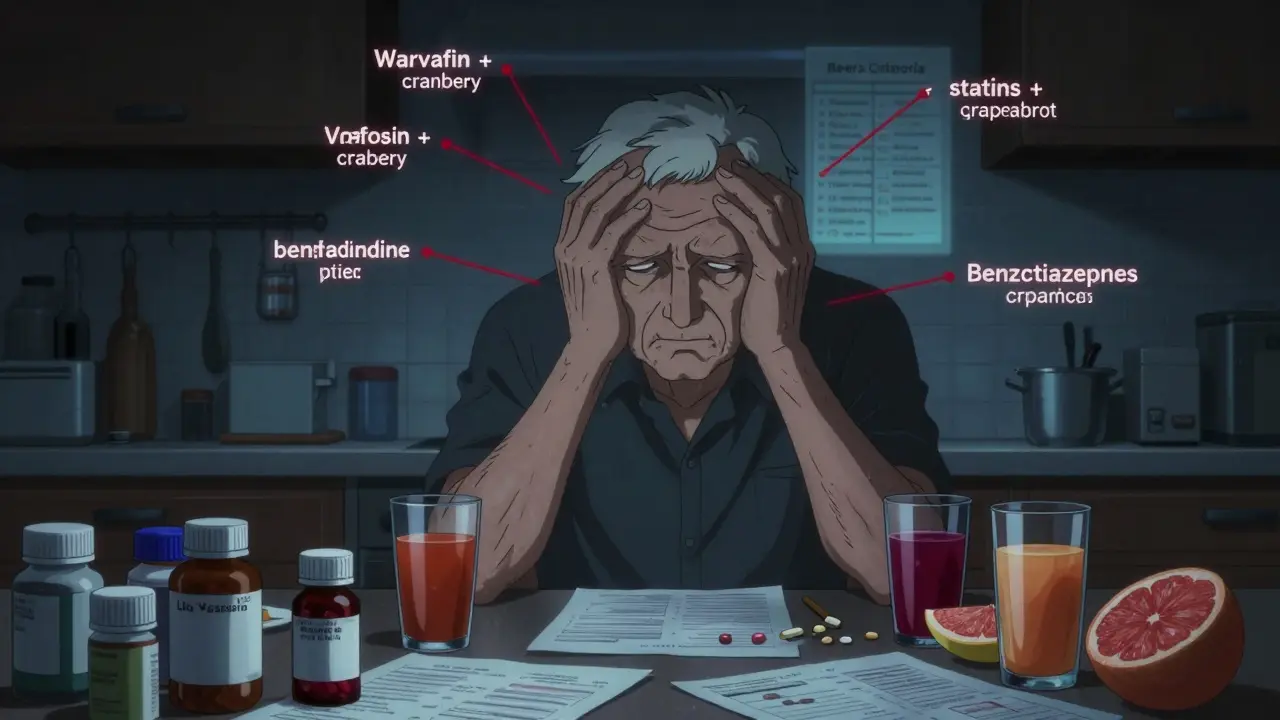
Polypharmacy - taking five or more medications - puts older adults at high risk of dangerous drug interactions, falls, and hospitalizations. This checklist helps you identify risky combinations and take control of your meds before it's too late.

Thiazolidinediones like pioglitazone and rosiglitazone help control blood sugar but carry a serious risk of fluid retention that can worsen heart failure. Learn who should avoid them and what safer alternatives exist.
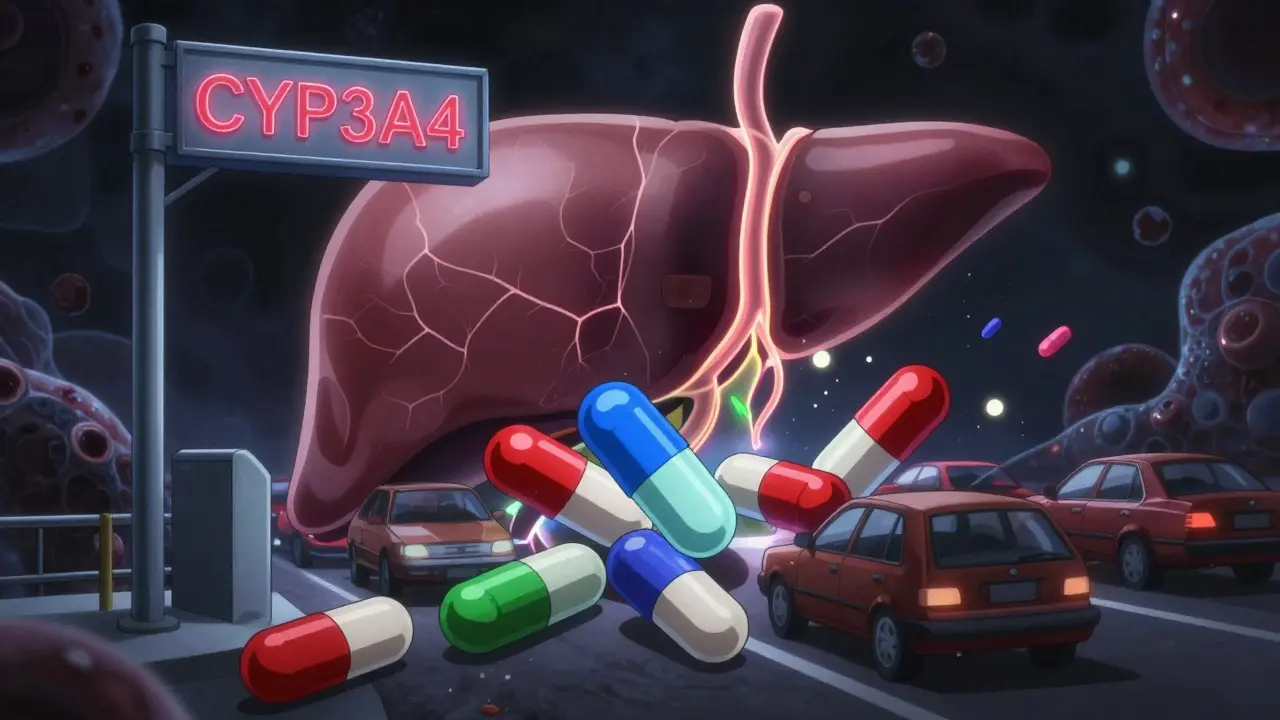
CYP450 enzymes process 90% of medications. When drugs compete for the same enzyme, levels can spike or crash - causing toxicity or treatment failure. Learn how common meds like statins, antidepressants, and antibiotics interact - and how to stay safe.

Learn how to correctly calculate power and sample size for bioequivalence studies to avoid costly failures. Understand key factors like CV%, GMR, and regulatory requirements for FDA and EMA compliance.